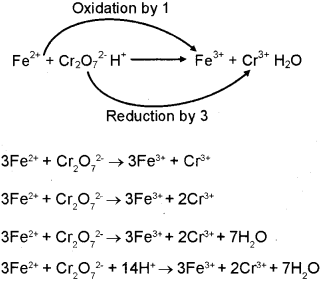
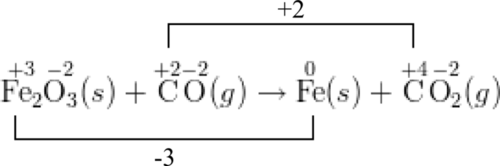
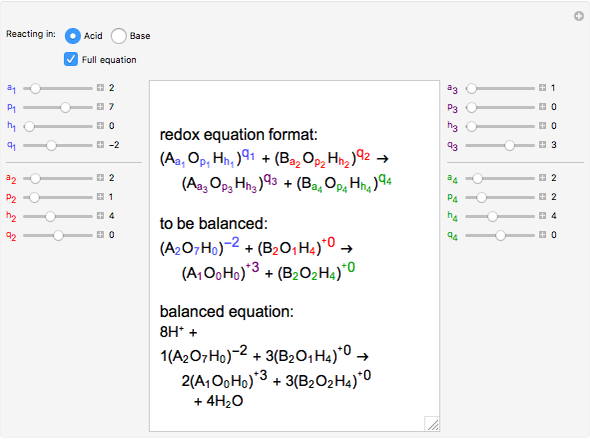
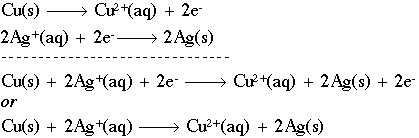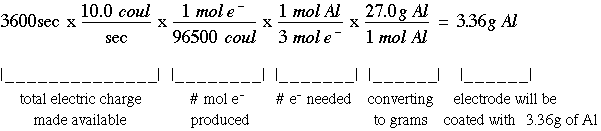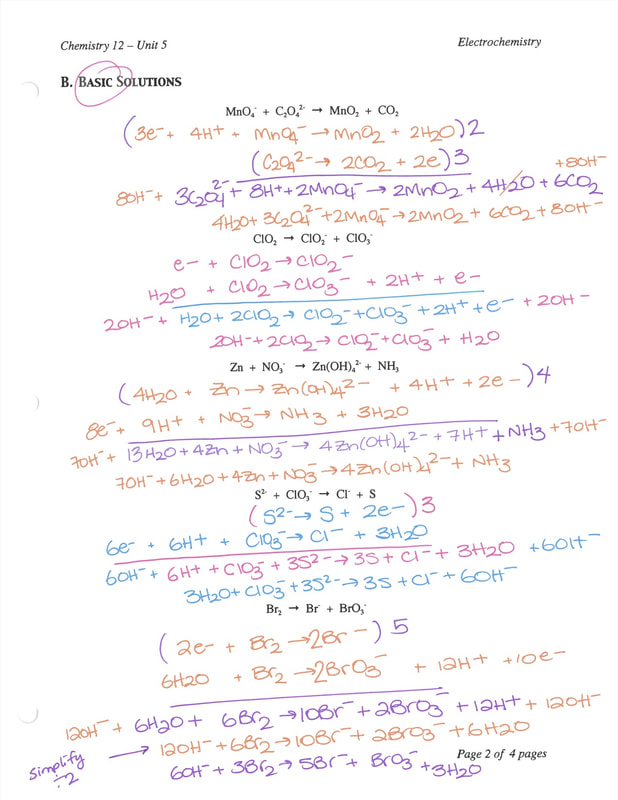Calculate the equilibrium constant for the redox reaction at 25°C. Sr(s) + Mg^(2+) → Sr^(2+)(aq) + Mg(s), - Sarthaks eConnect | Largest Online Education Community

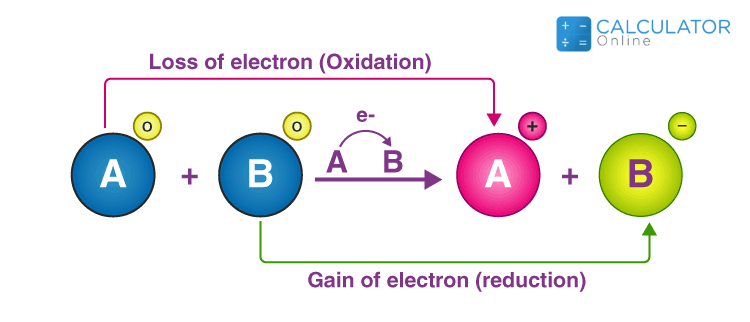
Oxidation and Reduction reactions- Definition, Reactions, Examples, Balancing the oxidation and reduction reactions, Videos and FAQs