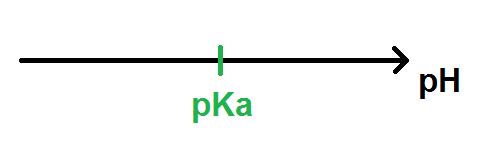
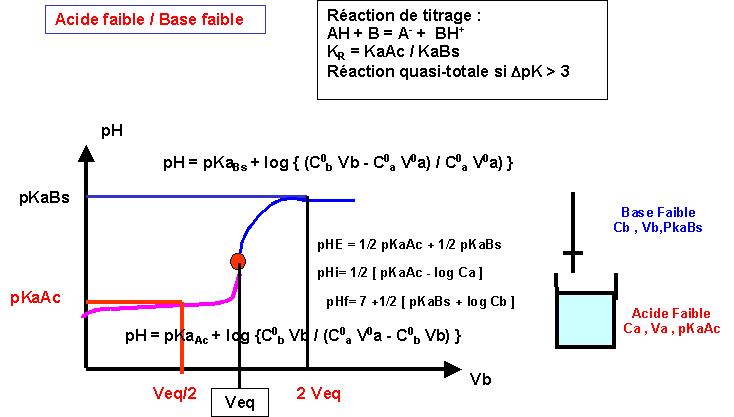
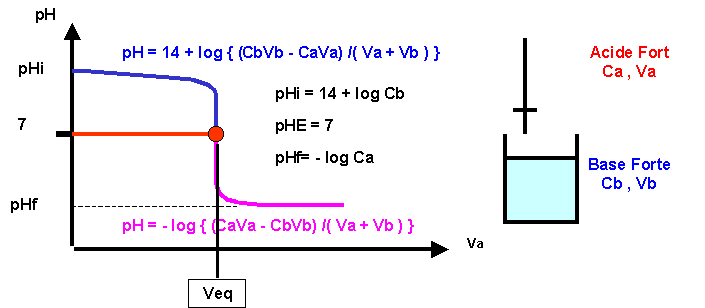
Validity of the Criterion pKa = pH at the Half Equivalence Point for the Potentiometric Evaluation of the Ionization Constant of
![Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube](https://i.ytimg.com/vi/SLPu7qlUdEA/maxresdefault.jpg)
Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube
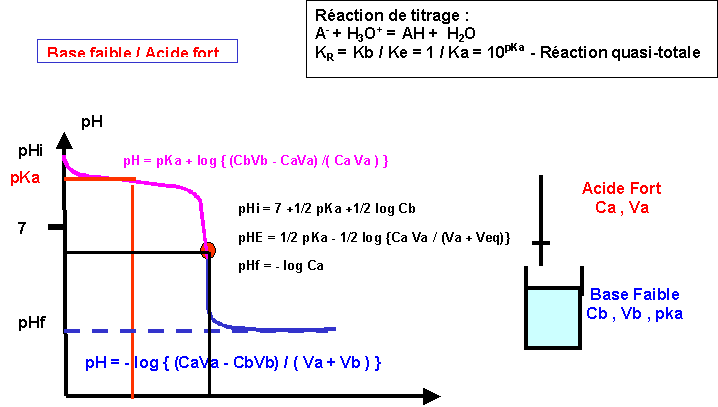
LES REACTIONS ACIDE-BASE 1. Le pH 1.1 Définition Le pH (potentiel hydrogène) est une grandeur qui donne une information sur la




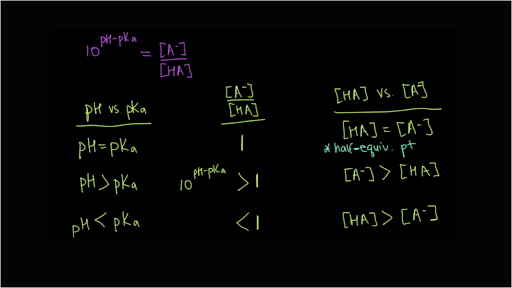
![cours TS / chimie / pH = pKa+log[A-]/[AH] , calcul de [A-]/[AH - YouTube cours TS / chimie / pH = pKa+log[A-]/[AH] , calcul de [A-]/[AH - YouTube](https://i.ytimg.com/vi/9uSKHUjti2s/hqdefault.jpg)


![Terminale spécialité : chapitre acide/base : démonstration pH = pKa + log ([A-]/(HA) - YouTube Terminale spécialité : chapitre acide/base : démonstration pH = pKa + log ([A-]/(HA) - YouTube](https://i.ytimg.com/vi/VqdraG_x93w/sddefault.jpg)

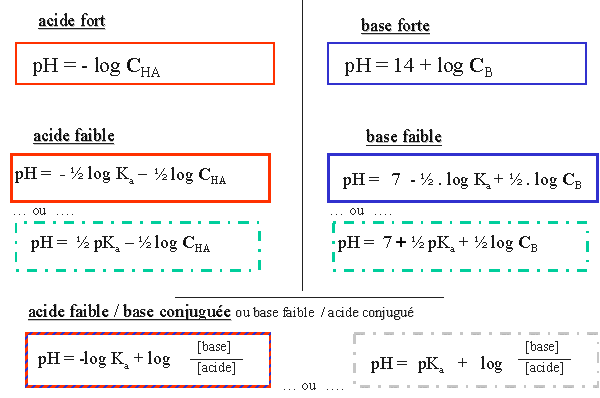
![Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube](https://i.ytimg.com/vi/2jpB30LsT8g/maxresdefault.jpg)