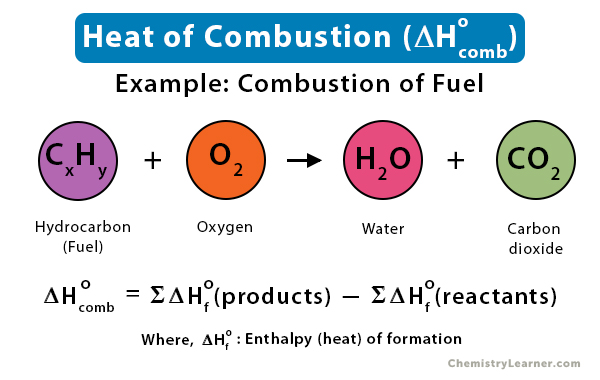
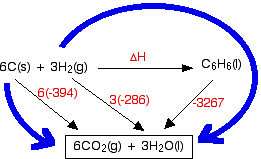
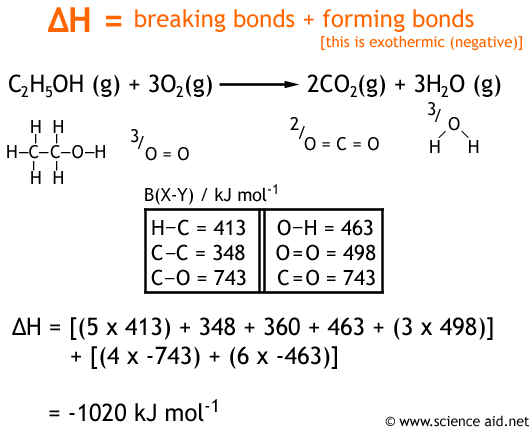
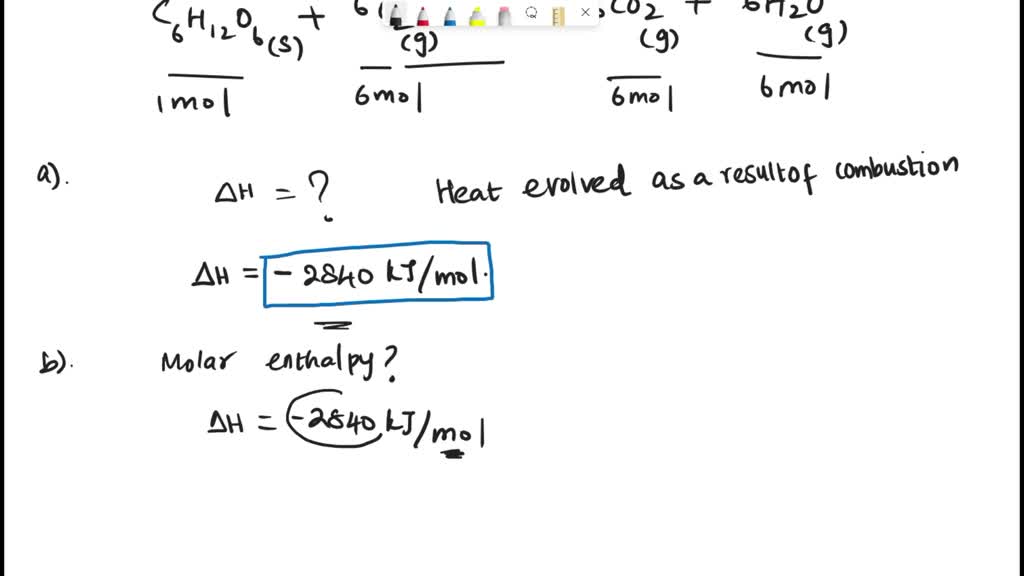⚗️Use the information from the diagram to calculate the enthalpy of combustion for methane.(IGNORE MY - Brainly.com

OneClass: The standard enthalpy of combustion of solid phenol (C6H50H) is-1 -1 -1 - 3054 kJ mol at 2...

Question Video: Determining the Standard Enthalpy of Formation of Ethanol Using Standard Enthalpies of Combustion | Nagwa
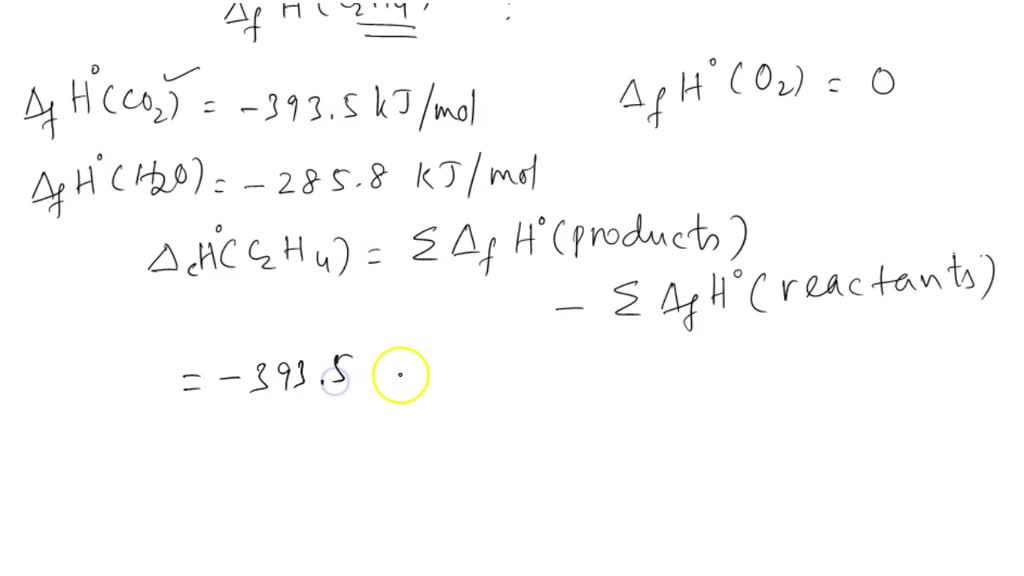
SOLVED: The standard enthalpy of combustion of ethene gas, C2H4 (g), is -1411.1 kJ/mol at 298 K. Given the following enthalpies of formation, calculate ΔHfo for C2H4 (g) CO2 (g): -393.5 kJ/mol

Question Video: Calculating Standard Enthalpy of Combustion of Methane Using Standard Enthalpies of Formation of Methane and Carbon Dioxide | Nagwa
21. Calculate the standard enthalpy of formation of n butane, given that the standard enthalpies of combustion of n butane, C(graphite) and H2(g) are –2878.5 kJ mol–1, –393.5 kJ mol–1 and –285.8