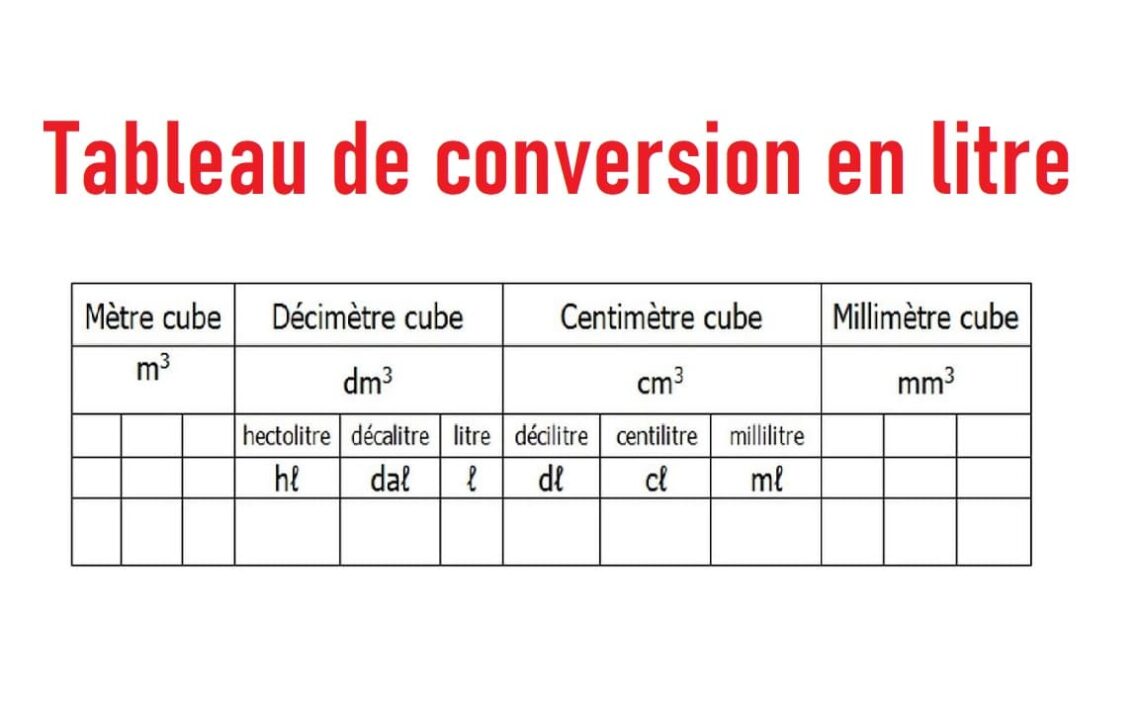
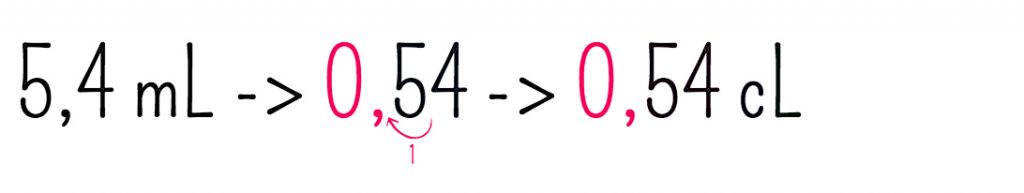SOLVED: A 450.0 -mL sample of a 0.257-M solution of silver nitrate is mixed with 400.0 mL of 0.200M calcium chloride. What is the concentration of Cl^- in solution after the reaction

SOLVED: What mass of silver chloride can be prepared by the reaction of 190.0 mL of 0.30 M silver nitrate with 160.0 mL of 0.14 M calcium chloride? Calculate the concentrations of